NMR
Measurement principle
Nuclear magnetic resonance (NMR) is a technique that directly measures atomic nuclei. Under the influence of a strong magnetic field pulse, the nuclear spins experience energy level splitting. By applying a radio frequency pulse of a specific frequency, the nuclear spins are excited to undergo energy level transitions, resulting in resonance signals.
The sample is mounted inside an Helmholtz whose dimension is dependent on the sample size and also the inductance that fits the experimental frequency. The coil is then installed on the sample probe, and the latter is inserted into the dewar which can generate the low-temperature environment. When target temperature is reached, proper pulsed magnet field is applied to perform the NMR measurements.
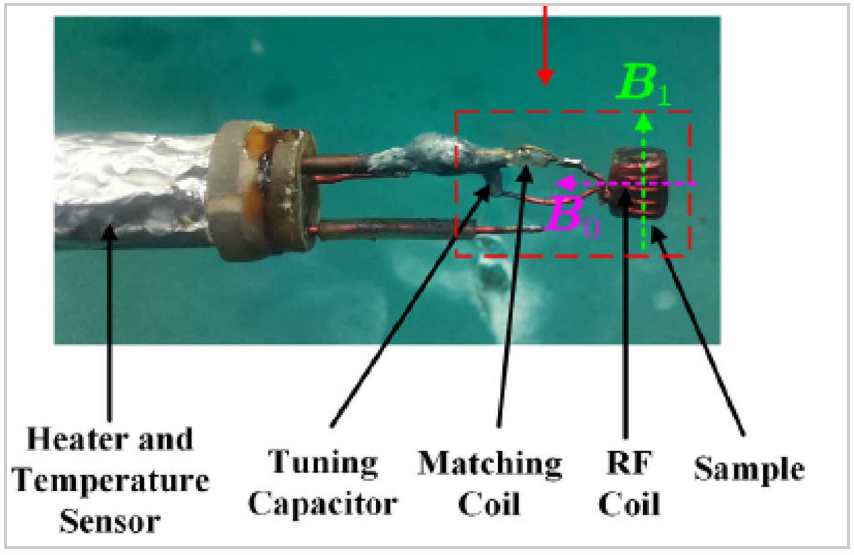
Fig.1 home-made NMR probe for low-temperature
Measurement Conditions
Magnetic Field: 0-60 T
Temperature: 2-300 K
Accuracy: Chemical/Knight shift precision less than 100-1000 ppm
Sample Morphology: powder, single crystal
Sample dimension: mm
Solid- / liquid-state NMR
Measurement principle
Nuclear magnetic resonance (NMR) is a technique that directly measures atomic nuclei. Under the influence of a strong magnetic field pulse, the nuclear spins experience energy level splitting. By applying a radio frequency pulse of a specific frequency, the nuclear spins are excited to undergo energy level transitions, resulting in resonance signals.
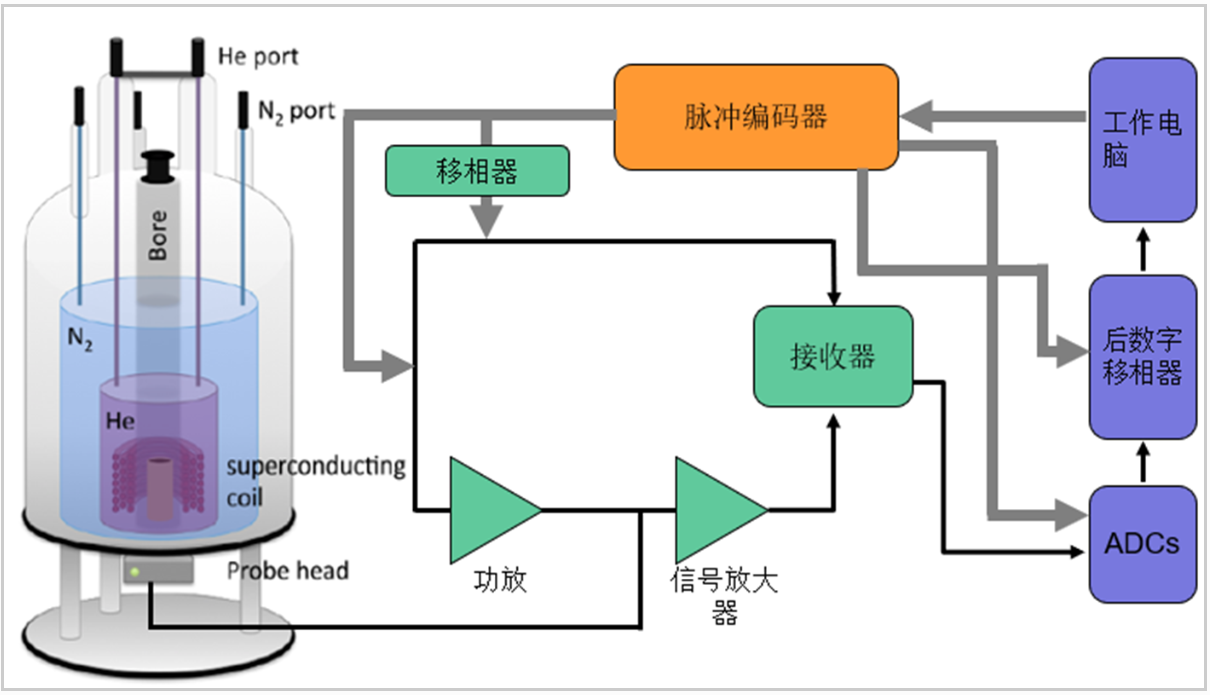
Fig.2 Schematic Diagram of Solid-State Steady-State NMR Device
Measurement Conditions
Magnetic Field Strength: 9.4T (1H 400 MHz), 11.7T (1H 500 MHz), 0-16T (Variable Field NMR)
Temperature: 200-1300K (Hot Probe), 1.5-300K (Cold Probe), 0.05-2K (Ultra-Low Temperature)
Measurement Accuracy: Pulse Sequence Control Accuracy not less than 100ns, Chemical/Knight Shift Measurement Accuracy not less than 1ppm
Sample Morphology: Powder, Single Crystal, Liquid
Sample Size: Volume not exceeding f4mm´5mm
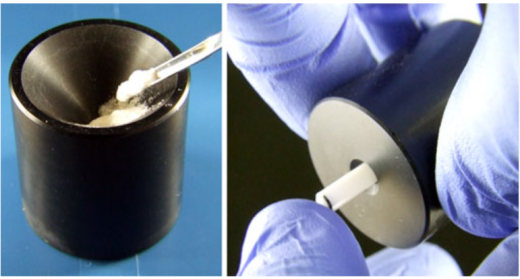
Fig.3 Solid-State NMR Experiment Sample Assembly Diagram

Fig.4 shows the 500 MHz solid state NMR.

Fig.5 shows the commercial MAS probe.
Nuclear magnetic resonance (NMR) instruments can precisely measure the Knight (chemical) shifts, nuclear quadrupole frequencies, and spin relaxation rates of solid materials under steady-state strong magnetic fields. This capability allows for the capture of the microscopic structure and dynamic evolution of materials, aiding in solving cutting-edge scientific issues such as the structure of chemical synthesis products, short-range order in amorphous glasses, liquid dynamics, and phase transitions.
The specific technical parameters are as follows: The maximum resonance frequencies for NMR measurements are 400 MHz (9.4T) and 500 MHz (11.7T). The excitation pulse power is 0-300W for the 1H channel and 0-300W for the X channel. The NMR spectrometers are equipped with commercial MAS probes of 2.5mm and 4mm. The maximum rotation speed of the 4mm probe is 10kHz, and the maximum rotation speed of the 2.5mm probe is 25kHz. The temperature measurement range of the commercial probes is from -80°C to 80°C. The magnetic field homogeneity is ±0.05ppm, and it is capable of measuring experiments such as chemical shift/Knight shift, T1/T2 relaxation times, and CPMAS cross-polarization.
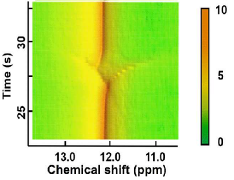
Fig.6 Pseudo 2D-NMR Data of Melting S-ibuprofen [J. Phys. Chem. Lett. 14, 9740, (2023)]
Key Contacts
Yongkang Luo Shiyu Liu
Email:mpzslyk#hust.edu.cn hustlsy#hust.edu.cn (Please replace # with @)
